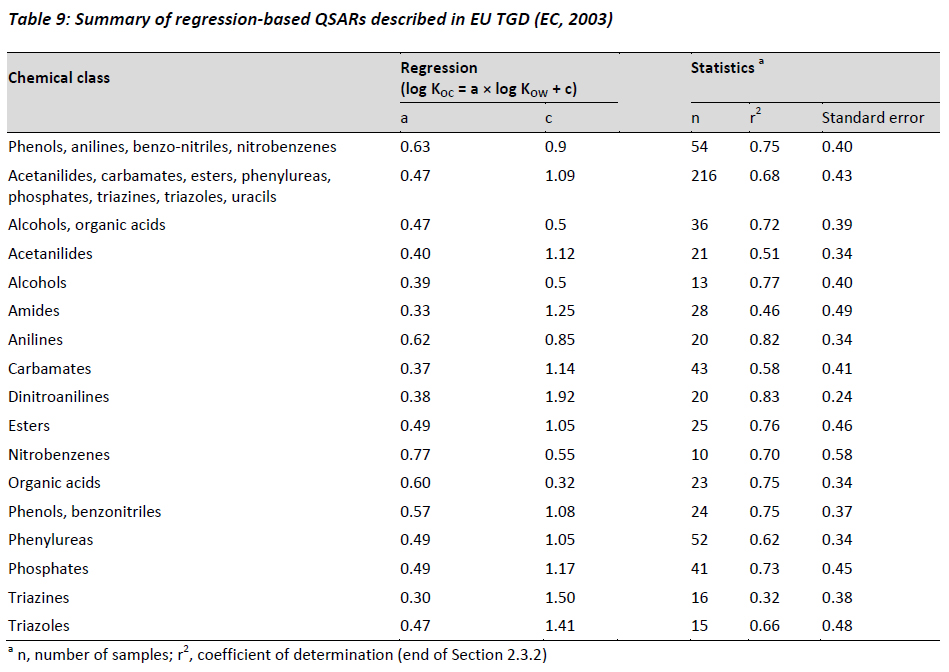
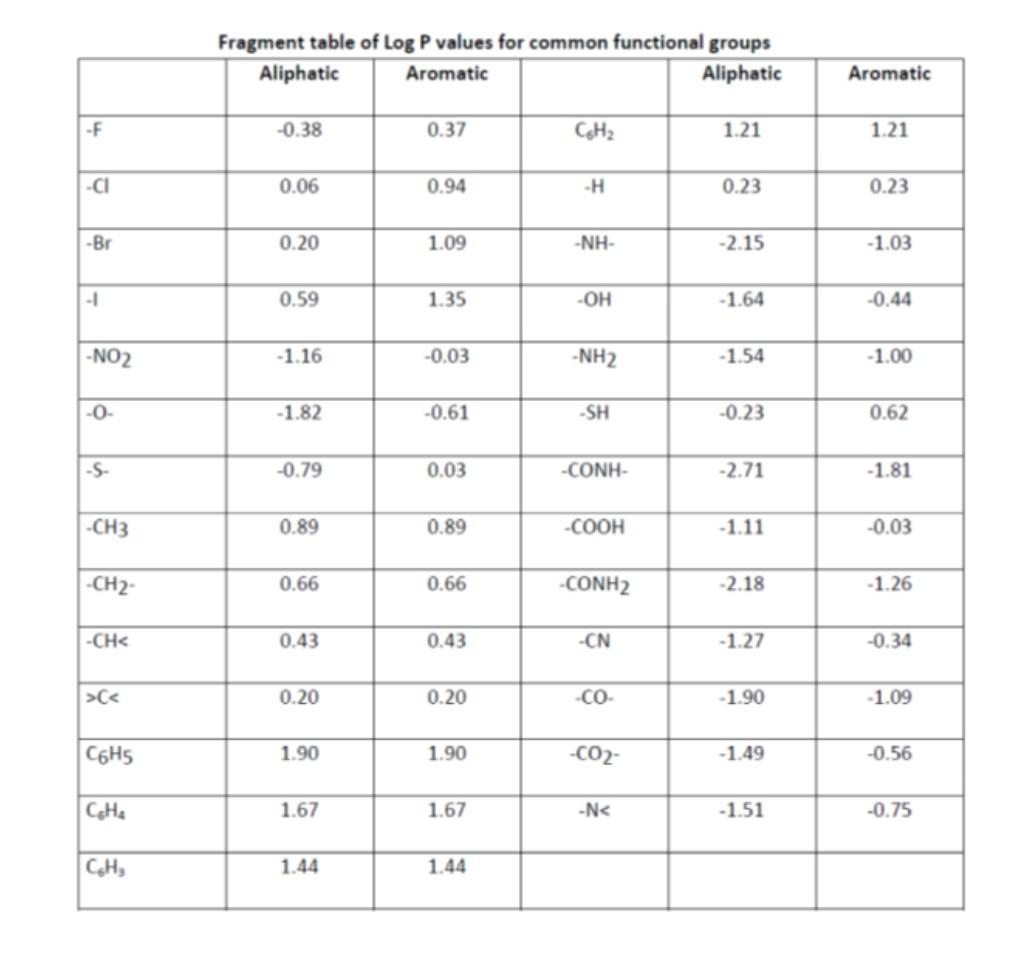
Table 3 from Determination of octanol‐water partitioning coefficients (KOW) of 61 organophosphorus and carbamate insecticides and their relationship to respective water solubility (S) values | Semantic Scholar
Modeling and prediction of octanol/water partition coefficient of pesticides using QSPR methods | Emerald Insight

Modeling the octanol–water partition coefficient of substituted phenols by the use of structure information - Jäntschi - 2007 - International Journal of Quantum Chemistry - Wiley Online Library
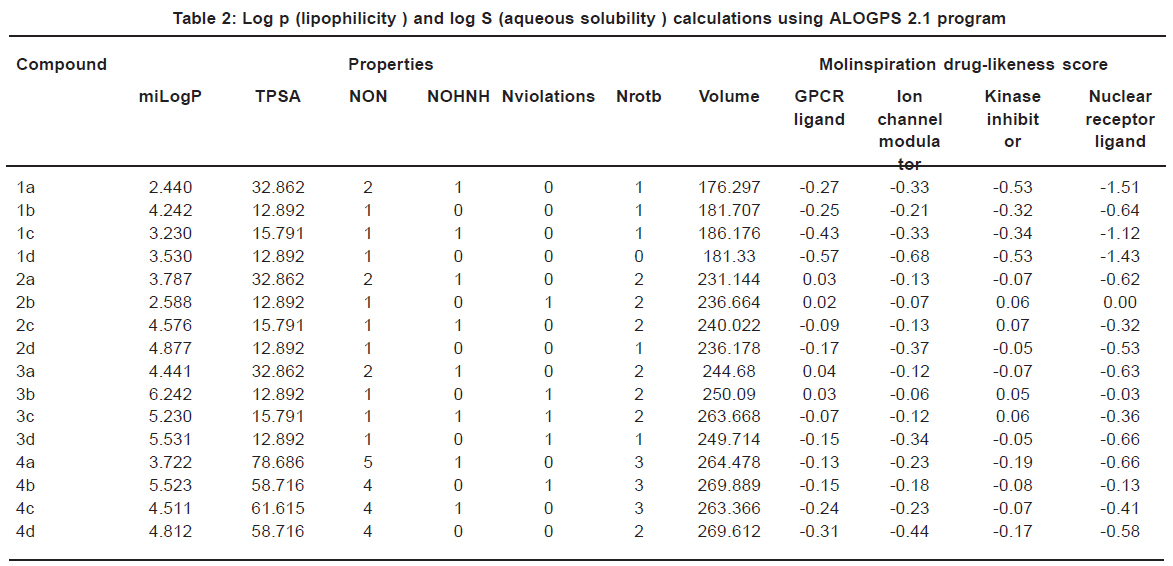
Calculation of Molecular Lipophilicity and Drug Likeness for Few Heterocycles : Oriental Journal of Chemistry
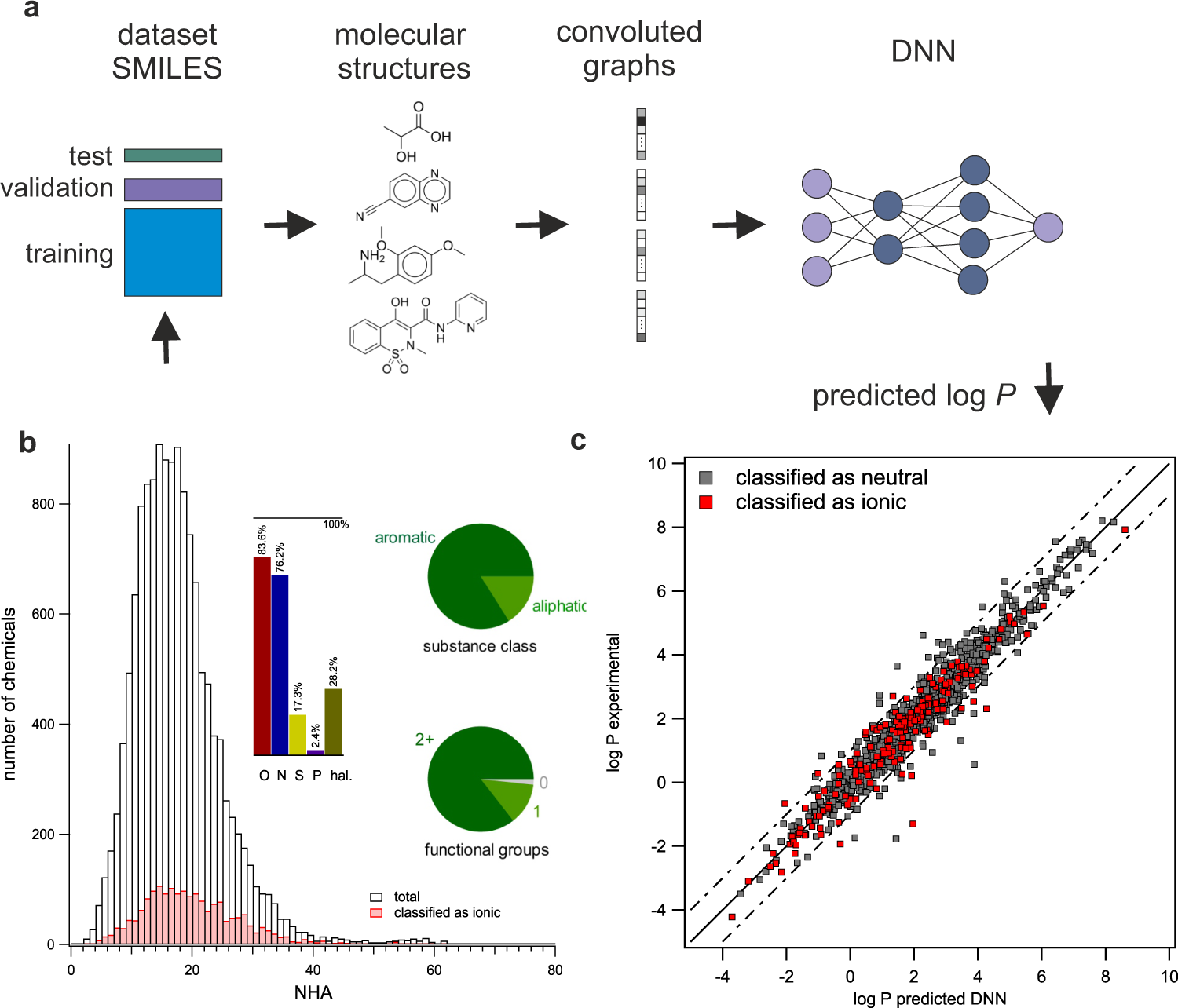
Exploring the octanol–water partition coefficient dataset using deep learning techniques and data augmentation | Communications Chemistry
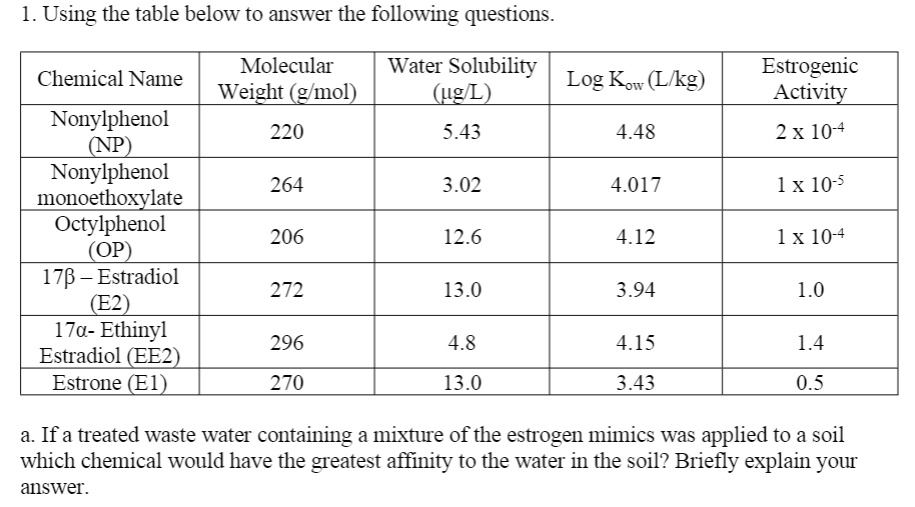
SOLVED: Using the table below to answer the following questions: Molecular Weight (g/mol) Water Solubility Log Kow (Likg) (ug/L) Nonylphenol (NP) 220 5.43 4.48 Nonylphenol monoethoxylate 264 3.02 4.017 Octylphenol (OP) 178

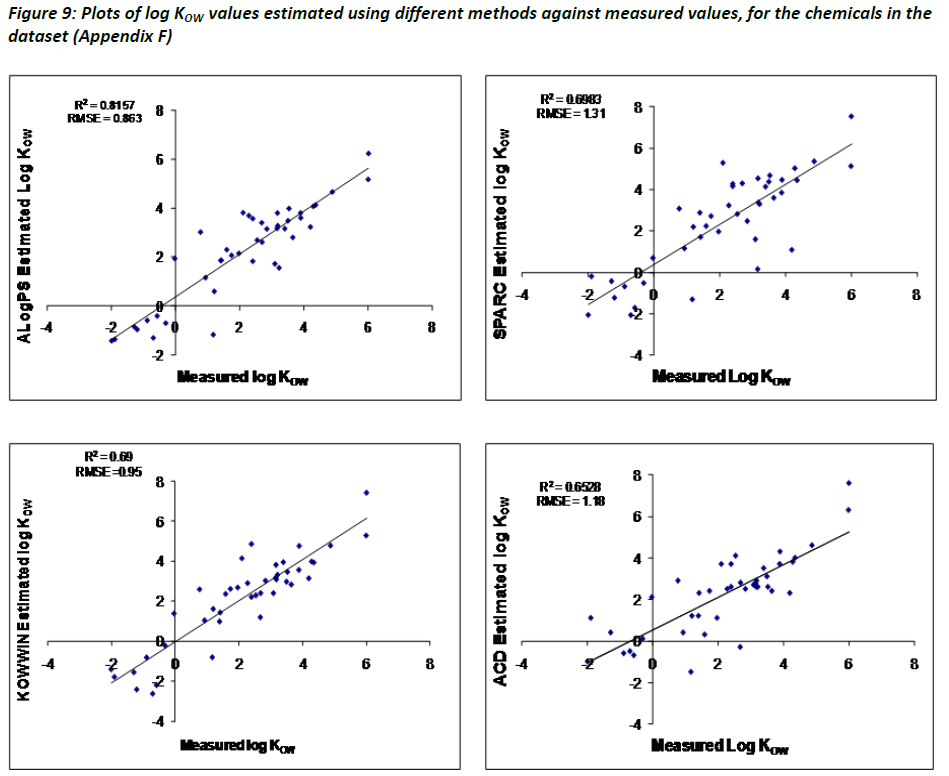
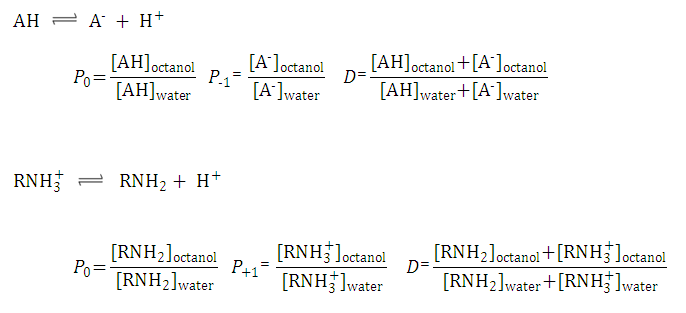
Evaluation of QSAR models for predicting the partition coefficient (log P) of chemicals under the REACH regulation - ScienceDirect

Table 2 from Determination of octanol‐water partitioning coefficients (KOW) of 61 organophosphorus and carbamate insecticides and their relationship to respective water solubility (S) values | Semantic Scholar

Table 1 from Acute toxicity and n-octanol/water partition coefficients of substituted thiophenols: determination and QSAR analysis. | Semantic Scholar
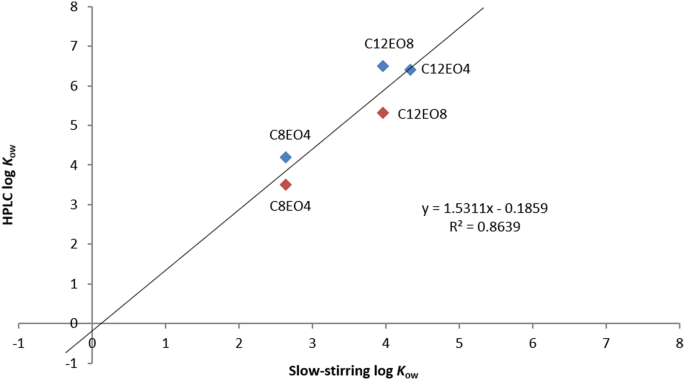
A comparison of log Kow (n-octanol–water partition coefficient) values for non-ionic, anionic, cationic and amphoteric surfactants determined using predictions and experimental methods | Environmental Sciences Europe | Full Text
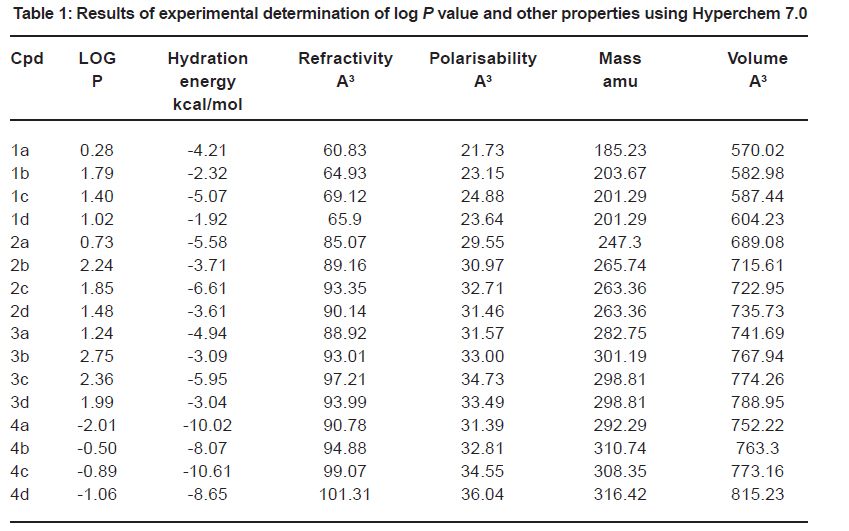
Calculation of Molecular Lipophilicity and Drug Likeness for Few Heterocycles : Oriental Journal of Chemistry

Table 1 from Determination of octanol‐water partitioning coefficients (KOW) of 61 organophosphorus and carbamate insecticides and their relationship to respective water solubility (S) values | Semantic Scholar

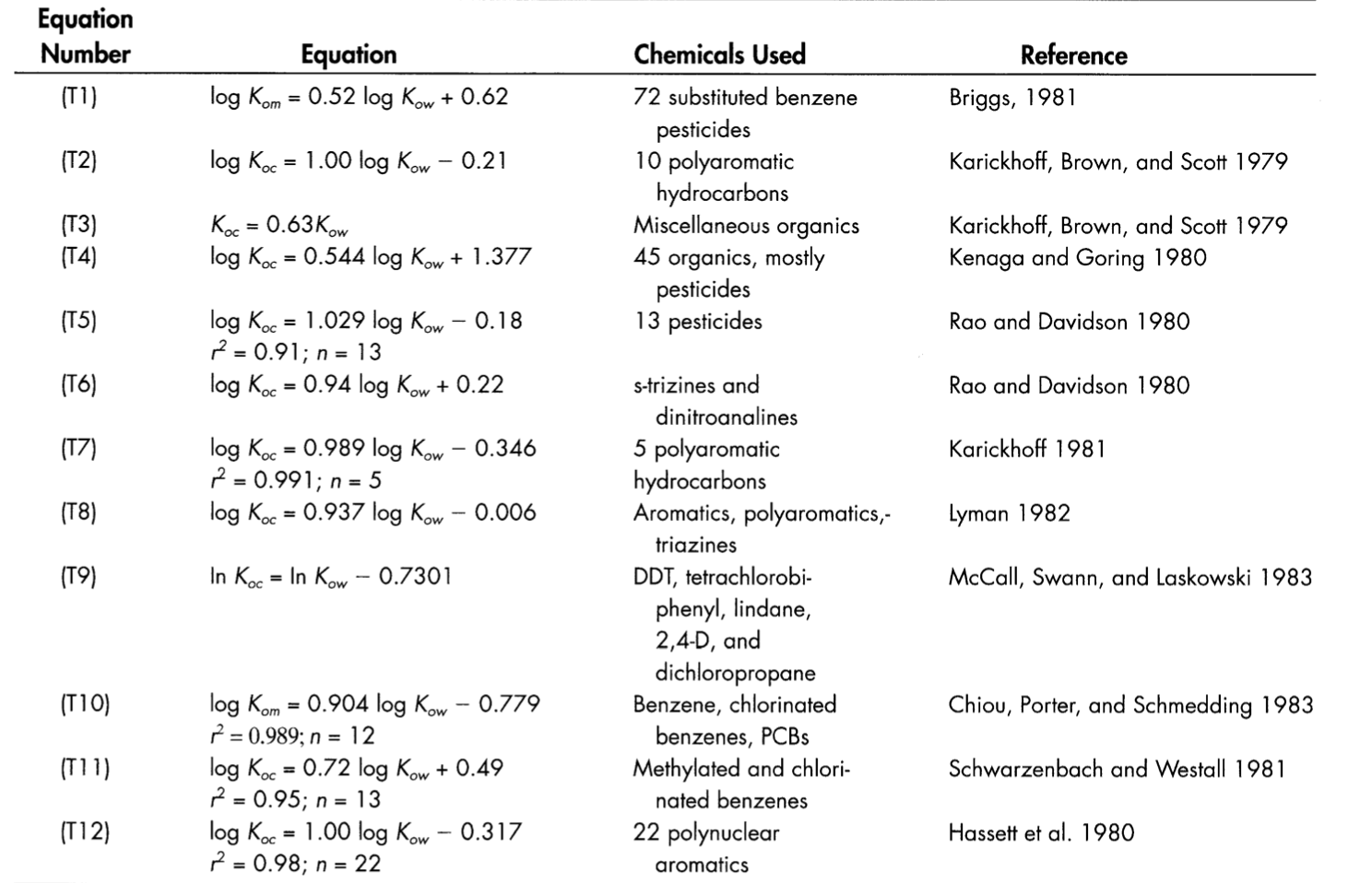
Table 5 from The search for reliable aqueous solubility (Sw) and octanol-water partition coefficient (Kow) data for hydrophobic organic compounds; DDT and DDE as a case study | Semantic Scholar